Want to know more about Tivdak?
Talk to a representative to get access to helpful resources, reimbursement programs, and more detailed information about Tivdak.
INDICATION: TIVDAK® is indicated for the treatment of adult patients with recurrent or metastatic cervical cancer (r/mCC) with disease progression on or after chemotherapy.
Talk to a representative to get access to helpful resources, reimbursement programs, and more detailed information about Tivdak.
Select the resources you would like to send via email
0 selected
Send by email

Prescribing Information
A user-friendly version of the Tivdak Prescribing Information


Tivdak Eye Care Provider Guide
A detailed brochure about Tivdak Premedication and Required Eye Care for your patients while on treatment


Dosing, Administration, and Eye Care Guide
A helpful guide to understanding and administering Tivdak


Eye Care Consult Form
A form to streamline communication between the eye care provider and the oncologist
Peer-reviewed publications


Efficacy and safety of tisotumab vedotin in innovaTV 204
This article, published by The Lancet Oncology in April 2021, reviews the clinical data from the innovaTV 204/GOG-3023/ENGOT-cx6 trial evaluating Tivdak in patients with previously treated recurrent or metastatic cervical cancer.
Select the resources you would like to send via email
0 selected
Send by email

Patient Brochure
A comprehensive guide for your patients, which includes an overview of Tivdak and what to expect from treatment


Caregiver Brochure
A handbook about caring for loved ones taking Tivdak


TivdakTexts Brochure
A guide to a text message support program designed to assist your patients throughout treatment


Important Facts
A brief summary of key information about Tivdak for your patients
An eye care provider will help monitor your patient’s eye health throughout treatment. Use this locator tool to find one.
TivdakTexts
Some benefits of TivdakTexts include:
Infusion reminders
Eye drop reminders
Inspirational messages
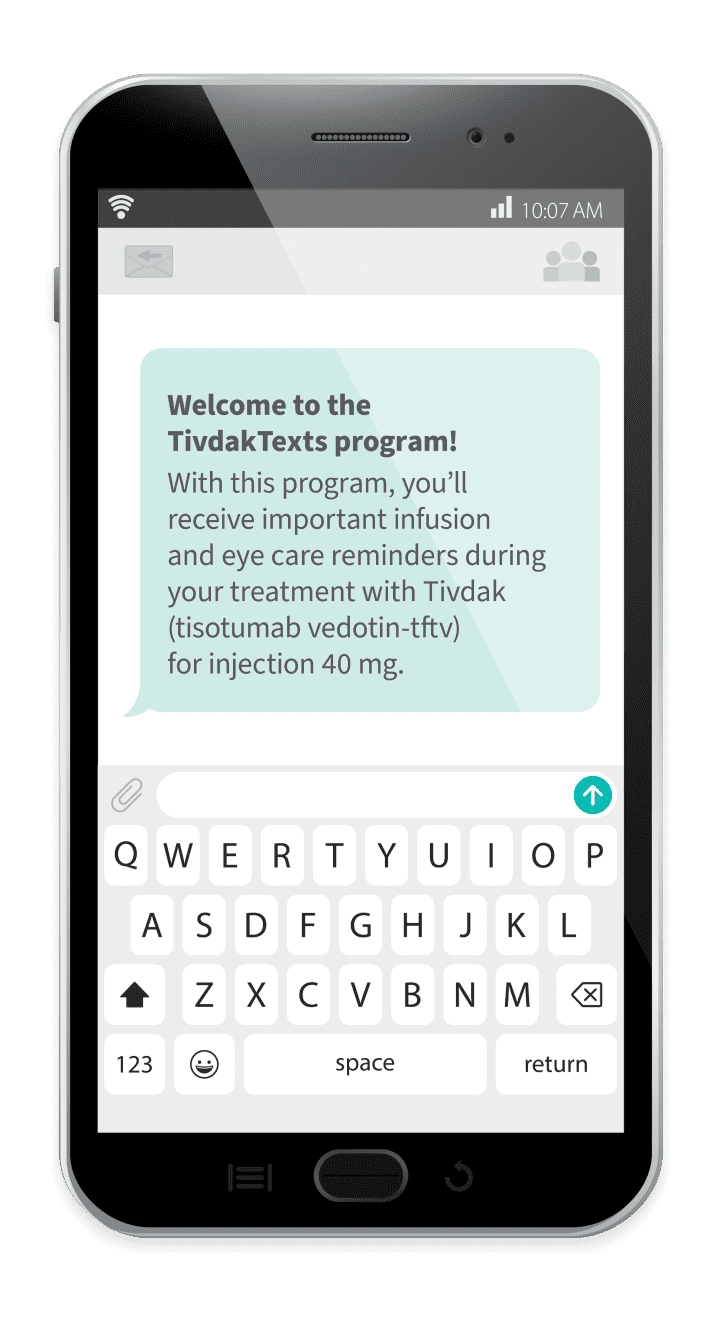
To sign up for TivdakTexts, patients can follow these steps:
On their mobile phone, they should text the word JOIN to 74277
When they get a message back, they should reply with the word AGREE to confirm their participation in the support program
They should then provide the following information:
Message and data rates may apply. Check with your mobile service provider. Message frequency will be based on your selections. Text HELP for help with the program. Text STOP to stop receiving messages. See Terms of Use and Conditions. By initiating a text message, you agree to receive automated text messages from Pfizer. You can unsubscribe at any time. You represent that you are the subscriber of the number provided and 18 or older. You must notify Pfizer immediately if you relinquish the number provided. Consent is not required as a condition of purchasing any products or services.
Seagen Secure®
Seagen Secure® is an access support program for people prescribed Tivdak. Oncology Access Advocates are an all-in-one resource to help your patients and their caregivers answer access-related questions throughout their treatment. Seagen Secure® offers information regarding coverage, financial assistance, and patient support to help patients access Tivdak.

Call 855-4SECURE (855-473-2873) or
Call 855-4SECURE (855-473-2873) or
visit seagensecure.com for more information.
*Seagen does not guarantee that enrollment will result in patient assistance, coverage, and/or reimbursement.
Get an in-depth look at the Tivdak mechanism of action
See phase 3 clinical trial results

Get to know the Tivdak infusion cycle
TIVDAK can cause severe ocular toxicities resulting in changes in vision, including severe vision loss, and corneal ulceration. Conduct an ophthalmic exam, including an assessment of ocular symptoms, visual acuity, and slit lamp exam of the anterior segment of the eye prior to initiation of TIVDAK, prior to every cycle for the first nine cycles, and as clinically indicated. Adhere to the required premedication and eye care before, during, and after infusion. Withhold TIVDAK until improvement and resume, reduce the dose, or permanently discontinue, based on severity.
Ocular adverse reactions: TIVDAK can cause severe ocular adverse reactions, including conjunctivitis, keratopathy (keratitis, punctate keratitis, and ulcerative keratitis), and dry eye (increased lacrimation, eye pain, eye discharge, pruritus, irritation, and foreign body sensation), that may lead to changes in vision and/or corneal ulceration.
Ocular adverse reactions occurred in 55% of patients with cervical cancer treated with TIVDAK across clinical trials. The most common were conjunctivitis (32%), dry eye (24%), keratopathy (17%), and blepharitis (5%). Grade 3 ocular adverse reactions occurred in 3.3% of patients, including severe ulcerative keratitis in 1.2% of patients. Nine patients (2.1%) experienced ulcerative keratitis (including one with perforation requiring corneal transplantation), six (1.4%) conjunctival ulcer, four (0.9%) corneal erosion, two (0.5%) conjunctival erosion, and two (0.5%) symblepharon.
In innovaTV 301, 8 patients (3.2%) experienced delayed ocular adverse reactions occurring more than 30 days after discontinuation of TIVDAK. These adverse reactions included 3 patients with ulcerative keratitis, and one patient (each) with keratitis, punctate keratitis and corneal erosion, blepharitis and conjunctival hyperemia, conjunctival scar, and conjunctivitis and xerophthalmia.
Refer patients to an eye care provider to conduct an ophthalmic exam prior to initiation of TIVDAK, prior to every cycle for the first nine cycles, and as clinically indicated. The exam should include visual acuity, slit lamp exam of the anterior segment of the eye, and an assessment of normal eye movement and ocular signs or symptoms which include dry or irritated eyes, eye secretions, or blurry vision.
Adhere to the required premedication and eye care before, during, and after infusion to reduce the risk of ocular adverse reactions. Monitor for ocular toxicity and promptly refer patients to an eye care provider for any new or worsening ocular signs and symptoms. Withhold, reduce, or permanently discontinue TIVDAK based on the severity or persistence of the ocular adverse reaction.
Peripheral neuropathy (PN) occurred in 39% of cervical cancer patients treated with TIVDAK across clinical trials; 6% of patients experienced Grade 3 PN. PN adverse reactions included peripheral sensory neuropathy (23%), PN (5%), paresthesia (3.8%), peripheral sensorimotor neuropathy (3.3%), muscular weakness (2.8%), and peripheral motor neuropathy (2.4%). One patient with another tumor type treated with TIVDAK at the recommended dose developed Guillain-Barre syndrome.
Monitor patients for signs and symptoms of neuropathy such as paresthesia, tingling or a burning sensation, neuropathic pain, muscle weakness, or dysesthesia. For new or worsening PN, withhold, then dose reduce, or permanently discontinue TIVDAK based on the severity of PN.
Hemorrhage occurred in 51% of cervical cancer patients treated with TIVDAK across clinical trials. The most common all grade hemorrhage adverse reaction was epistaxis (33%). Grade 3 hemorrhage occurred in 4% of patients.
Monitor patients for signs and symptoms of hemorrhage. For patients experiencing pulmonary or central nervous system hemorrhage, permanently discontinue TIVDAK. For Grade ≥2 hemorrhage in any other location, withhold until bleeding has resolved, blood hemoglobin is stable, there is no bleeding diathesis that could increase the risk of continuing therapy, and there is no anatomical or pathologic condition that can increase the risk of hemorrhage recurrence. After resolution, either resume treatment or permanently discontinue TIVDAK.
Pneumonitis that is severe, life-threatening, or fatal can occur in patients treated with antibody-drug conjugates containing vedotin, including TIVDAK. Among cervical cancer patients treated with TIVDAK across clinical trials, 4 patients (0.9%) experienced pneumonitis, including 1 patient who had a fatal outcome.
Monitor patients for pulmonary symptoms of pneumonitis. Symptoms may include hypoxia, cough, dyspnea or interstitial infiltrates on radiologic exams. Infectious, neoplastic, and other causes for such symptoms should be excluded through appropriate investigations. Withhold TIVDAK for patients who develop persistent or recurrent Grade 2 pneumonitis and consider dose reduction. Permanently discontinue TIVDAK in all patients with Grade 3 or 4 pneumonitis.
Severe cutaneous adverse reactions (SCAR), including events of fatal or life-threatening Stevens-Johnson syndrome (SJS), can occur in patients treated with TIVDAK. SCAR occurred in 1.6% of cervical cancer patients treated with TIVDAK across clinical trials. Grade ≥3 SCAR occurred in 0.5% of patients, including 1 patient who had a fatal outcome.
Monitor patients for signs or symptoms of SCAR, which include target lesions, worsening skin reactions, blistering or peeling of the skin, painful sores in mouth, nose, throat, or genital area, fever or flu-like symptoms, and swollen lymph nodes. If signs or symptoms of SCAR occur, withhold TIVDAK until the etiology of the reaction has been determined. Early consultation with a specialist is recommended to ensure greater diagnostic accuracy and appropriate management. Permanently discontinue TIVDAK for confirmed Grade 3 or 4 SCAR, including SJS.
Embryo-fetal toxicity: TIVDAK can cause fetal harm when administered to a pregnant woman. Advise patients of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with TIVDAK and for 2 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with TIVDAK and for 4 months after the last dose.
Across clinical trials of TIVDAK in 425 patients with r/mCC, the most common (≥25%) adverse reactions, including laboratory abnormalities, were hemoglobin decreased (45%), PN (39%), conjunctival adverse reactions (38%), nausea (37%), fatigue (36%), aspartate aminotransferase increased (33%), epistaxis (33%), alopecia (31%), alanine aminotransferase increased (30%), and hemorrhage (28%).
innovaTV 301 Study: 250 patients with r/mCC with disease progression on or after systemic therapy
Serious adverse reactions occurred in 33% of patients receiving TIVDAK; the most common (≥2%) were urinary tract infection (4.8%), small intestinal obstruction (2.4%), sepsis, abdominal pain, and hemorrhage (each 2%). Fatal adverse reactions occurred in 1.6% of patients who received TIVDAK, including acute kidney injury, pneumonia, sepsis, and SJS (each 0.4%).
Adverse reactions leading to permanent discontinuation occurred in 15% of patients receiving TIVDAK; the most common (≥3%) were PN and ocular adverse reactions (each 6%). Adverse reactions leading to dose interruption occurred in 39% of patients receiving TIVDAK; the most common (≥3%) were ocular adverse reactions (16%) and PN (6%). Adverse reactions leading to dose reduction occurred in 30% of patients receiving TIVDAK; the most common (≥3%) were PN and ocular adverse reactions (each 10%). The ocular adverse reactions included conjunctival disorders (4.8%), keratopathy (4%), and dry eye (0.8%).
innovaTV 204 Study: 101 patients with r/mCC with disease progression on or after chemotherapy
Serious adverse reactions occurred in 43% of patients; the most common (≥3%) were ileus (6%), hemorrhage (5%), pneumonia (4%), PN, sepsis, constipation, and pyrexia (each 3%). Fatal adverse reactions occurred in 4% of patients who received TIVDAK, including septic shock, pneumonitis, sudden death, and multisystem organ failure (each 1%).
Adverse reactions leading to permanent discontinuation occurred in 13% of patients receiving TIVDAK; the most common (≥3%) were PN (5%) and corneal adverse reactions (4%). Adverse reactions leading to dose interruption occurred in 47% of patients; the most common (≥3%) were PN (8%), conjunctival adverse reactions, and hemorrhage (each 4%). Adverse reactions leading to dose reduction occurred in 23% of patients; the most common (≥3%) were conjunctival adverse reactions (9%) and corneal adverse reactions (8%).
Strong CYP3A4 inhibitors: Concomitant use with strong CYP3A4 inhibitors may increase unconjugated monomethyl auristatin E (MMAE) exposure, which may increase the risk of TIVDAK adverse reactions. Closely monitor patients for TIVDAK adverse reactions.
Moderate or severe hepatic impairment: MMAE exposure and adverse reactions are increased. Avoid use.
Lactation: Advise lactating women not to breastfeed during TIVDAK treatment and for at least 3 weeks after the last dose.
Please see full Prescribing Information, including BOXED WARNING for TIVDAK.
TIVDAK is indicated for the treatment of adult patients with recurrent or metastatic cervical cancer (r/mCC) with disease progression on or after chemotherapy.
Reference: 1. TIVDAK [Prescribing Information]. Bothell, WA: Seagen Inc. April 2024.
TIVDAK can cause severe ocular toxicities resulting in changes in vision, including severe vision loss, and corneal ulceration. Conduct an ophthalmic exam, including an assessment of ocular symptoms, visual acuity, and slit lamp exam of the anterior segment of the eye prior to initiation of TIVDAK, prior to every cycle for the first nine cycles, and as clinically indicated. Adhere to the required premedication and eye care before, during, and after infusion. Withhold TIVDAK until improvement and resume, reduce the dose, or permanently discontinue, based on severity.
Ocular adverse reactions: TIVDAK can cause severe ocular adverse reactions, including conjunctivitis, keratopathy (keratitis, punctate keratitis, and ulcerative keratitis), and dry eye (increased lacrimation, eye pain, eye discharge, pruritus, irritation, and foreign body sensation), that may lead to changes in vision and/or corneal ulceration.
Ocular adverse reactions occurred in 55% of patients with cervical cancer treated with TIVDAK across clinical trials. The most common were conjunctivitis (32%), dry eye (24%), keratopathy (17%), and blepharitis (5%). Grade 3 ocular adverse reactions occurred in 3.3% of patients, including severe ulcerative keratitis in 1.2% of patients. Nine patients (2.1%) experienced ulcerative keratitis (including one with perforation requiring corneal transplantation), six (1.4%) conjunctival ulcer, four (0.9%) corneal erosion, two (0.5%) conjunctival erosion, and two (0.5%) symblepharon.
In innovaTV 301, 8 patients (3.2%) experienced delayed ocular adverse reactions occurring more than 30 days after discontinuation of TIVDAK. These adverse reactions included 3 patients with ulcerative keratitis, and one patient (each) with keratitis, punctate keratitis and corneal erosion, blepharitis and conjunctival hyperemia, conjunctival scar, and conjunctivitis and xerophthalmia.
Refer patients to an eye care provider to conduct an ophthalmic exam prior to initiation of TIVDAK, prior to every cycle for the first nine cycles, and as clinically indicated. The exam should include visual acuity, slit lamp exam of the anterior segment of the eye, and an assessment of normal eye movement and ocular signs or symptoms which include dry or irritated eyes, eye secretions, or blurry vision.
Adhere to the required premedication and eye care before, during, and after infusion to reduce the risk of ocular adverse reactions. Monitor for ocular toxicity and promptly refer patients to an eye care provider for any new or worsening ocular signs and symptoms. Withhold, reduce, or permanently discontinue TIVDAK based on the severity or persistence of the ocular adverse reaction.
Peripheral neuropathy (PN) occurred in 39% of cervical cancer patients treated with TIVDAK across clinical trials; 6% of patients experienced Grade 3 PN. PN adverse reactions included peripheral sensory neuropathy (23%), PN (5%), paresthesia (3.8%), peripheral sensorimotor neuropathy (3.3%), muscular weakness (2.8%), and peripheral motor neuropathy (2.4%). One patient with another tumor type treated with TIVDAK at the recommended dose developed Guillain-Barre syndrome.
Monitor patients for signs and symptoms of neuropathy such as paresthesia, tingling or a burning sensation, neuropathic pain, muscle weakness, or dysesthesia. For new or worsening PN, withhold, then dose reduce, or permanently discontinue TIVDAK based on the severity of PN.
Hemorrhage occurred in 51% of cervical cancer patients treated with TIVDAK across clinical trials. The most common all grade hemorrhage adverse reaction was epistaxis (33%). Grade 3 hemorrhage occurred in 4% of patients.
Monitor patients for signs and symptoms of hemorrhage. For patients experiencing pulmonary or central nervous system hemorrhage, permanently discontinue TIVDAK. For Grade ≥2 hemorrhage in any other location, withhold until bleeding has resolved, blood hemoglobin is stable, there is no bleeding diathesis that could increase the risk of continuing therapy, and there is no anatomical or pathologic condition that can increase the risk of hemorrhage recurrence. After resolution, either resume treatment or permanently discontinue TIVDAK.
Pneumonitis that is severe, life-threatening, or fatal can occur in patients treated with antibody-drug conjugates containing vedotin, including TIVDAK. Among cervical cancer patients treated with TIVDAK across clinical trials, 4 patients (0.9%) experienced pneumonitis, including 1 patient who had a fatal outcome.
Monitor patients for pulmonary symptoms of pneumonitis. Symptoms may include hypoxia, cough, dyspnea or interstitial infiltrates on radiologic exams. Infectious, neoplastic, and other causes for such symptoms should be excluded through appropriate investigations. Withhold TIVDAK for patients who develop persistent or recurrent Grade 2 pneumonitis and consider dose reduction. Permanently discontinue TIVDAK in all patients with Grade 3 or 4 pneumonitis.
Severe cutaneous adverse reactions (SCAR), including events of fatal or life-threatening Stevens-Johnson syndrome (SJS), can occur in patients treated with TIVDAK. SCAR occurred in 1.6% of cervical cancer patients treated with TIVDAK across clinical trials. Grade ≥3 SCAR occurred in 0.5% of patients, including 1 patient who had a fatal outcome.
Monitor patients for signs or symptoms of SCAR, which include target lesions, worsening skin reactions, blistering or peeling of the skin, painful sores in mouth, nose, throat, or genital area, fever or flu-like symptoms, and swollen lymph nodes. If signs or symptoms of SCAR occur, withhold TIVDAK until the etiology of the reaction has been determined. Early consultation with a specialist is recommended to ensure greater diagnostic accuracy and appropriate management. Permanently discontinue TIVDAK for confirmed Grade 3 or 4 SCAR, including SJS.
Embryo-fetal toxicity: TIVDAK can cause fetal harm when administered to a pregnant woman. Advise patients of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with TIVDAK and for 2 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with TIVDAK and for 4 months after the last dose.
Across clinical trials of TIVDAK in 425 patients with r/mCC, the most common (≥25%) adverse reactions, including laboratory abnormalities, were hemoglobin decreased (45%), PN (39%), conjunctival adverse reactions (38%), nausea (37%), fatigue (36%), aspartate aminotransferase increased (33%), epistaxis (33%), alopecia (31%), alanine aminotransferase increased (30%), and hemorrhage (28%).
innovaTV 301 Study: 250 patients with r/mCC with disease progression on or after systemic therapy
Serious adverse reactions occurred in 33% of patients receiving TIVDAK; the most common (≥2%) were urinary tract infection (4.8%), small intestinal obstruction (2.4%), sepsis, abdominal pain, and hemorrhage (each 2%). Fatal adverse reactions occurred in 1.6% of patients who received TIVDAK, including acute kidney injury, pneumonia, sepsis, and SJS (each 0.4%).
Adverse reactions leading to permanent discontinuation occurred in 15% of patients receiving TIVDAK; the most common (≥3%) were PN and ocular adverse reactions (each 6%). Adverse reactions leading to dose interruption occurred in 39% of patients receiving TIVDAK; the most common (≥3%) were ocular adverse reactions (16%) and PN (6%). Adverse reactions leading to dose reduction occurred in 30% of patients receiving TIVDAK; the most common (≥3%) were PN and ocular adverse reactions (each 10%). The ocular adverse reactions included conjunctival disorders (4.8%), keratopathy (4%), and dry eye (0.8%).
innovaTV 204 Study: 101 patients with r/mCC with disease progression on or after chemotherapy
Serious adverse reactions occurred in 43% of patients; the most common (≥3%) were ileus (6%), hemorrhage (5%), pneumonia (4%), PN, sepsis, constipation, and pyrexia (each 3%). Fatal adverse reactions occurred in 4% of patients who received TIVDAK, including septic shock, pneumonitis, sudden death, and multisystem organ failure (each 1%).
Adverse reactions leading to permanent discontinuation occurred in 13% of patients receiving TIVDAK; the most common (≥3%) were PN (5%) and corneal adverse reactions (4%). Adverse reactions leading to dose interruption occurred in 47% of patients; the most common (≥3%) were PN (8%), conjunctival adverse reactions, and hemorrhage (each 4%). Adverse reactions leading to dose reduction occurred in 23% of patients; the most common (≥3%) were conjunctival adverse reactions (9%) and corneal adverse reactions (8%).
Strong CYP3A4 inhibitors: Concomitant use with strong CYP3A4 inhibitors may increase unconjugated monomethyl auristatin E (MMAE) exposure, which may increase the risk of TIVDAK adverse reactions. Closely monitor patients for TIVDAK adverse reactions.
Moderate or severe hepatic impairment: MMAE exposure and adverse reactions are increased. Avoid use.
Lactation: Advise lactating women not to breastfeed during TIVDAK treatment and for at least 3 weeks after the last dose.
Please see full Prescribing Information, including BOXED WARNING for TIVDAK.
TIVDAK is indicated for the treatment of adult patients with recurrent or metastatic cervical cancer (r/mCC) with disease progression on or after chemotherapy.
Fill out your information below and we will reach out to answer your questions.
All fields required unless indicated optional.
for requesting information!
By clicking this link, you will be redirected to a website that is neither owned nor controlled by Pfizer. Pfizer is not responsible for the content or services of this site.
Please select 1 or more resources.